You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Introduction
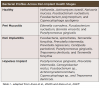
Peri-implant mucositis is characterized by inflammation of the adjacent mucosa, bleeding upon probing, and the absence of supportive bone loss.2,4 Peri-implantitis, conversely, manifests with similar indications of inflammation and bleeding upon probing, accompanied with observable radiographic bone loss and deeper pocket depths.2,4 It is important to acknowledge that mucositis is reversible. However, if left untreated, it may progress to peri-implantitis, which can advance more swiftly than periodontitis in natural dentition, ultimately resulting in implant failure.2,4 Moderate to severe peri-implantitis and related bone loss mainly occur in the mandibular and maxillary incisor/canine regions. In contrast, peri-mucositis often impacts the maxillary molar, maxillary incisor/canine, and mandibular premolar locations.5 Hence, understanding these patterns and accurately identifying the first and subtle clinical and radiographic indicators of peri-implant mucositis and peri-implantitis is imperative to reduce the incidence and prevent disease development.
Microbiota Around Implant Sites
The examination of oral microbiota in pathogenic contexts is essential, particularly with the advancement of implantology. Identifying causes of pathology and developing solutions can help patients maintain the aesthetic and biomechanical functionality of implants, which can otherwise be compromised by biofilm accumulation and the subsequent loss of peri-implant bone support.5,6 Oral dysbiotic biofilm is recognized as the prevalent cause of periodontitis and peri-implantitis, as shown by their clinical manifestations and pathophysiology.5,6 In comparison to a healthy periodontium, both conditions are marked by the presence of Gram-negative anaerobic bacteria at the injury sites.5,6 Peri-implantitis displays increased microbial diversity, notably a higher prevalence of Fusobacterium spp. and Treponema spp., especially when compared with healthy implants sites.5,6,7
The colonization of anaerobic bacteria and the bacterial byproducts, such as lipopolysaccharides, adhere to the micro gap of dental implants, triggering an increased production of cytokines that inhibit bone formation while promoting osteoclastogenesis.2,6,7 The microbiome surrounding implants affected by mucositis, peri-implantitis, or diagnosed as unsavable differs significantly from that of natural dentition. Consequently, treatment strategies targeting bacteria predominantly associated with periodontitis may be less effective in managing implant-related conditions.6
Electrostatic attraction and mechanical bonding foster the initial adhesion of bacteria to the implant surface, followed by the release of extracellular polymers. As the biofilm phase progresses, bacteria quorum sensing-an intercellular signaling molecule-leads to the growth of co-aggregate communities capable of inciting an inflammatory response of the host tissue and continuation of constructing mature biofilm that becomes resistant to antimicrobial therapy.6,8,9 Furthermore, the oral microbiota is frequently inhabited by a variety of yeasts, including Candida albicans, C. guilliermondii, C. glabrata, Rhodotorula spp., and Trichosporon spp., which are essential for the formation of biofilms.6 This is a critical point to consider in the management of peri-implant diseases, as they are often resistant to antifungal therapies. Understanding the bacterial profiles at different stages of peri-implant health-from adhesion to dispersion-is essential for developing recommendations for patients, ranging from early preventive care to the management of advanced disease stages.
Implant Osseointegration
Earlier understanding of osseointegration presumed a direct structural and functional joining between living bone and the carrying-load implant surface. However, it is now understood that true osseointegration is achieved when there is no relative progressive movement between the implant and the surrounding bone.10 The physiochemical characteristics of the implant's outermost layer and its interaction with adjacent critical tissues influence the success or failure of osseointegration.11 Morphologically, dental and implant periodontal tissues exhibit several similarities, characterized by well-keratinized oral epithelium and a segment of connective tissue in indirect contact with the implant and tooth. Nevertheless, implanted connective tissue has an increased amount of collagen and a reduced number of fibroblasts.1
Innovations in Implant Design
Implants are designed as hydrophobic, with a smoother surface, or as hydrophilic, distinguished by roughened surfaces produced by sandblasting and etching. The latter demonstrates superior osseointegration and accelerated osseous growth.1 Hydrophilic implant surfaces exhibit an enhanced rate of osseointegration owing to their rapid interaction with the site clot. Upon surgical placement of the implant, fibrin production and platelet release occur as a protective mechanism to initiate wound healing, followed by clot reabsorption (fibrinolysis), osteoclastic activity, and the involvement of mesenchymal cells involved bone remodeling.1,10,11 Innovative methods for engineering titanium surfaces with unique meso, micro, and nanoscale roughness have shown enhanced osteoconductive and osseointegrative qualities.8
Supplementary biomaterial treatments applied to the titanium implant surface have further enhanced the facilitation of a good bone response and biomechanical performance and can build a robust and enduring epithelial biological seal essential for preventing bacterial contamination.1,8 Some compounds used or tested for coating titanium implants include polyhydroxyalkanoates, calcium phosphate, carbon, bisphosphonates, hydroxyapatite, bone-stimulating chemicals, bioactive glass, bioactive ceramics, collagen, chitosan, titanium alloys, and fluoride.1,8 Calcium phosphate (CaP) coated implants have been shown to enhance the first healing phase of osseointegration. Fluoride on the titanium surface interacts with hydroxyapatite in bone tissue to form fluorapatite, subsequently promoting osteoblast proliferation. Copper-containing titanium alloys have shown prolonged antibacterial action.1,8
Another innovative approach under research is the use of bifunctional peptides to combat bacterial colonization and biofilm formation, reducing the adverse host inflammatory response that destroys the surrounding implant tissue. A bifunctional peptide film may be used on new implants or reapplied to existing implants to mitigate bacterial colonization.12 These bifunctional peptides have been shown to achieve nearly full surface coverage within minutes, allowing for a quick chairside application time of existing implants, resilience to brushing with manual or electric toothbrushes and maintaining antimicrobial activity following bacterial challenges. In one study, researchers developed a unique chemical peptide by combining two parts: one derived from a natural human protein called β-defensin-3, which kills bacteria, and another that helps the peptide stick to titanium. These parts were connected using a triple glycine sequence.13 The modified titanium surfaces were tested and found to effectively kill harmful oral bacteria, including Streptococcus gordonii, Fusobacterium nucleatum, and Porphyromonas gingivalis.13
Management Strategies to Reduce Implant Failure
Management of peri-implant infections to preserve an implant necessitates antibiotic medication, which may elevate the patient's risk of antibiotic resistance, discomfort, and financial burden.9 As such, titanium implants must possess lasting antibacterial qualities to enhance initial osseointegration and ensure sustained success by inhibiting the formation of bacterial biofilms on their surface.1,8 One approach to preventatively managing and reducing microorganisms' adhesion to dental implant surfaces is to incorporate special coatings that resist or prevent adhesion. Implants coated with antifouling polymer coatings or nano topographical patterns have shown to effectively prevent or restrict bacterial adhesion for extended periods and mitigate the adverse effects on the surrounding tissue.8
Plaque index scores should be assessed regularly to monitor patient's effectiveness of plaque control.9 Patients with dental implants must maintain an effective home care regimen to support the health of the oral cavity and surrounding tissues.9,14,15Electric toothbrushes are recommended for daily oral hygiene, as they demonstrate superior efficacy in biofilm reduction and are linked to healthier implant sites when used for 2-3 minutes daily.15
Recent studies examining antiseptic mouth rinses, such as chlorhexidine, have not demonstrated benefits in reducing implant pocket depths, even as an adjunctive to debridement therapy. This highlights the need for further research to determine whether chlorhexidine provides any meaningful advantage in such applications.14,16,18 While chlorhexidine has demonstrated a reduction in bacterial load and inflammation, its effect on pocket depth reduction remains negligible in implant areas.7,16 Conversely, rinsing with 0.25% sodium hypochlorite as a standalone treatment has shown efficacy in decreasing pocket depths and bleeding of peri-implantitis sites.17 Furthermore, The American Dental Association Council recognizes 0.1% sodium hypochlorite as a viable antiseptic agent.17 Locally administered antibiotics can be more effective than systemic antibiotics alone in treating peri-implantitis and provide superior outcomes compared to chlorhexidine rinse.20 Minocycline microspheres or ointment, doxycycline gel, chlorhexidine chips, and tetracycline fibers have effectively decreased implant probing depth, enhanced attachment levels, and ameliorated peri-implantitis.19,20
New research of natural antibacterial polymers with antibacterial, antioxidant, and anti-inflammatory properties, such as chitin, an abundant natural amino polysaccharide, has shown promising results.2 Synthesized water-soluble quaternized chitin has proven effective in eliminating planktonic P. gingivalis and destroying established biofilms. Additionally, quaternized chitin has demonstrated the ability to inhibit the expression of typical inflammatory factors associated with P. gingivalis and reduce the production of reactive oxidative stress induced by P. gingivalis's virulence factor, lipopolysaccharide (LPS).2
Conclusion
As implantology expands and the number of dental patients seeking rehabilitation through implant therapy increases, oral health professionals must remain informed about the latest research to effectively cater to their patients and develop a genuinely personalized care strategy tailored to their specific requirements.