You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Halitosis, also commonly known as “bad breath,” is a concern of many patients seeking help from dental professionals. The prevalence of this condition affects approximately 50% of the adult population worldwide and has been reported as being a persistent chronic problem in 25% of the cases.1,2 While the etiology of the problem is occasionally indicative of a systemic medical condition that requires referral to a physician, 90% of cases of halitosis can be traced to local sources in the oral cavity.3 Currently, with the strong emphasis on the importance of personal image and interpersonal relationships in modern society, it is not surprising that patients seek assistance for what is considered to be an embarrassing ailment.
The word halitosis is derived from both Latin (halitus – breath) and Greek (osis – condition, action, or pathologic process) origins. Combining these roots in English translates to the commonly used term of “bad breath” to describe this condition.4
The consistent odorous breath associated with halitosis can be caused by a single factor or multiple etiologies, including: 1) poor oral hygiene; 2) deep carious lesions; 3) periodontal disease; 4) oral infections; 5) peri-implant disease; 6) pericoronitis; 7) mucosal ulcerations; 8) certain foods; 9) impacted food; 10) tongue coating; 11) poor salivary flow rate; and 12) improper cleaning of dentures; all of these have been implicated as etiologic factors.5 Interactions between oral bacterial species and these co-factors produce the chemical agents of malodor, which include volatile sulfide compounds (VSC), diamines, and short chain fatty acids.6 The VSC are considered the main components of malodor, with hydrogen sulfide (H2S), methyl mercaptan (CH3SH), and dimethylsulfide ((CH3)2S) being the main offenders. Hydrogen sulfide can be identified by its “rotten egg” like odor, and methyl mercaptan and dimethylsulfide have both been described as having a smell similar to “rotten cabbage.” These compounds form following the breakdown by anaerobic Gram-negative oral bacteria of sulfur-containing substrates in food debris, saliva, blood, and epithelial cells.7 With the many possible causes, it is advisable to evaluate each patient suffering from oral malodor with all the various etiologies in mind, arrive at a differential diagnosis, and recommend an individualized course of treatment.
The purpose of this review is to propose a diagnostic process that dentists and hygienists can use to determine the presence of halitosis, discuss the specific etiologies related to each patient’s problem, and recommend an individualized approach to treatment. Additionally, recommendations are made on when to refer a patient to a healthcare professional in other specialized fields.
Diagnosis and Assessment of Halitosis
The first step in assessment of a patient presenting with halitosis is to perform a thorough patient interview. A person’s overall systemic health, dental habits, and diet are all key factors that can contribute to the problem. Additionally, it is important to be aware of how long the patient has noticed, or been informed by others, of the presence of malodor. While there is currently no ideal standard test to measure the extent and cause of halitosis, there are several types of evaluations available. Direct methods include the practitioner directly sniffing the breath or patient plaque samples, as well as measurement of sulfur-containing substances by halimetry or gas chromatographic.7 Each of these methods has its advantages and disadvantages.
The simplest and most common direct test for oral malodor is the direct sniffing of expired air. This is known as an “organoleptic” or “hedonic” assessment.8 While there is a subjective nature to this method and it might not be pleasant for the dentist or hygienist performing the evaluation, it is most similar to having oral malodor detected in daily life. A scale ranging from 0 to 5 (0 = no odor, 5 = extremely foul odor) introduced by Rosenberg and colleagues is often used as the quantitative value in malodor evaluations.9 These results should be recorded to assess the degree of disease and to evaluate the success of treatment strategies. The patient’s own organoleptic evaluation of collected saliva samples can serve as motivation for compliance to the individualized treatment plan. The patient could also be asked to evaluate the odor of floss containing retrieved plaque samples immediately after flossing his or her own teeth. Having the patient acknowledge and describe the malodor present (and improvement after treatment) directly allows the patient to understand the need for intervention, his or her role in following the prescribed treatment plan, and the need for maintenance once improvement is observed.
An examiner planning to use an organoleptic method of assessment requires calibration. This process involves ensuring there is not a defect in the smelling capacity (anosmia) of the examiner. He or she must be able to smell and recognize different odors (qualitative assessment) and also be able to detect odors at low concentrations (quantitative assessment). The qualitative assessment can be completed via a commercially available “smell identification test,” which ensures that an examiner is able to differentiate smells at a level comparable to or better than the general population for his or her given age. The quantitative assessment can be evaluated by being able to detect decreasing concentrations of inexpensive organic components such as phenethyl alcohol, thiophene, or pyridine prepared by a pharmacist.10
An advantage of the organoleptic method of assessment is that it closely mimics daily life and is also inexpensive. A major disadvantage is the subjective nature of the evaluation. It is also possible that an examiner can detect a strong odor that he or she may feel classifies as malodor (eg, onions, garlic, alcohol, cigars) but which the individual patient does not consider offensive.
Odorous gases, specifically the aforementioned H2S, CH3SH, and (CH3)2S, can be readily detected using gas chromatography (GC). These volatile sulfur compounds compose about 90% of the VSC in the oral cavity.6 A disadvantage of GC is the relatively high cost and requirement of highly trained personnel for values to be properly detected and measured. Also, gas chromatographers are not portable, thus making a re-evaluation of malodor treatment less readily available in the office.1
A relatively inexpensive, portable industrial sulfide monitor has recently been sold commercially that measures VSC chairside with a suction pump and sensor. Studies using this monitor reported positive correlations between sulfide concentrations and ratings on the Rosenberg organoleptic assessment scale.11,12 A major disadvantage of this device is its inability to differentiate between various sulfides and other odorous gasses associated with halitosis, such as putrescine and cadaverine. In addition, false positives are common when the patient has recently eaten odorous food or performed oral hygiene procedures with distinct smelling toothpastes or mouthwashes.8-12
Etiology
Once the diagnosis of halitosis is made via any of the above methods, it is the dental practitioner’s task to determine the etiology of the malodor. An in-depth oral and radiographic evaluation are essential in determining if oral lesions, caries, periodontal disease, peri-implantitis, tongue coating, oral abscesses, or tonsillar crypts are contributing to the offensive smell.
Systemic conditions that can contribute to oral malodor include sinusitis, bronchitis, liver cirrhosis, kidney insufficiency, and diabetes.10,13,14 In addition to a complete dental examination, it is important that a patient receive an examination from his or her physician if any of these systemic conditions exist. Furthermore, some people continually insist they have bad breath when the presence of malodor is absent. Such people are considered to have a condition called “halitophobia.”10 Some of these patients also have additional psychopathological symptoms best managed by psychological professionals. This fear of having bad breath when in reality no oral malodor is present has been cited to effect 0.5% to 1% of the adult population.11
Treatment In the Dental Office
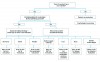
Once halitosis has been determined to be present, the etiology of the malodor must be identified prior to deciding on a treatment protocol (Figure 1). A combination of mechanical and chemical therapies has shown to be effective when treating halitosis.5 It is the dentist’s responsibility to advise the patient of lifestyle changes and hygiene practices that can aid in reducing the offending malodor. The individualized treatment regimen determined for each patient is a combined effort with professional care in the dental office, proper home care by the patient, and/or referral to a physician when necessary. A systematic determination of diagnosis involves considering each of the following etiological categories: dental, otolaryngeal, systemic, and/or social. Oftentimes the etiology is caused by more than one of these factors acting synergistically.
Dental Etiologies
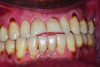
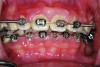
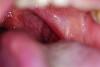
Clinical and radiographic examinations are essential to diagnose dental pathologies. Carious lesions, defective restorations, or fractured cusps, restorations or teeth (Figure 2 and Figure 3) can be sources of malodor both due to food impaction and foul odor associated with necrotic pulp tissue in cases of severe decay.10 Proper excavation, restorations, and regular follow-up are essential for the successful treatment of these patients. Areas of food impaction should be evaluated for restorations to close the embrasure space. Proper instruction of home care via dental floss and/or interproximal brushes is also required. An individual prone to caries as the source of malodor may also benefit from the adjunctive use of a fluoride-containing mouthrinse or toothpaste.
A rotten egg or cabbage smell is indicative of VSC being the main cause of halitosis. These compounds are usually associated with periodontal disease (Figure 3 and Figure 4) and/or tongue coating. Patients suffering from periodontitis or peri-implantitis require professional periodontal treatment in order to eliminate periodontal pathogens.5 Following a professional scaling and root planing appointment, Gram-negative anaerobic bacteria responsible for causing periodontal disease and producing VSC are reduced.5 This, in turn, encourages repopulation with Gram-positive cocci and bacterial rods that are associated with oral health.
Instruction for proper home care that includes brushing and use of interdental cleaning devices is an essential element to the management of halitosis. During the dental hygiene appointment, the dentist or hygienist should demonstrate a brushing regimen, such as the modified Bass technique, that enables sulcular penetration of the bristles.15 Demonstration of the use of floss to clean below the gingiva and interproximal brushes for larger embrasure spaces or under fixed bridges are additional aids for effective home plaque management. Ironically, many patients presenting with halitosis have little plaque and biofilm accumulations because of overzealous brusing and flossing habits that they use to try and cure the disease.
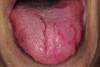
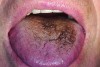
It has been shown that the dorsum of the tongue is the primary source of VSC.5 The anatomic structure of the tongue provides a large surface area with, in some cases, numerous fissures and grooves (Figure 5). This becomes an ideal niche for bacteria in the oral cavity (Figure 6). Although halitosis is usually associated with poor oral hygiene and periodontal conditions, evidence suggests that anaerobic microorganisms present in the tongue coating are often the cause of halitosis.11 Studies have shown a significant reduction of oral bacteria and the resulting VSC by patients who mechanically remove the biofilm from the teeth and tongue during daily oral hygiene. Tongue cleaning is more effective when a specifically designed tongue scraper is used instead of a toothbrush.16 Examples of the many effective scrapers include the BreathRx® Tongue Scraper (Philips Oral Healthcare, www.philipsoralhealthcare.com), the Butler G-U-M® Fresh-R Tongue Cleaner (Sunstar Americas, Inc., www.sunstar.com), and the Pureline Oralcare® Tongue Cleaner (Pureline Oralcare, www.purelineoralcare.com).
Denture wearers may be prone to halitosis because of the build-up of biofilm and food substrates under dentures. Failure to remove the denture and clean the edentulous areas, teeth supporting the partial denture, and/or the denture itself can cause halitosis (Figure 7). Proper care of dentures includes rinsing them after eating and scrubbing them daily with gentle dishwashing soap and soft-bristled denture brushes. Additionally, dentures should be soaked in water or mild commercially available denture cleaner and should not be worn overnight. Similar build-up of biofilm and food substrates can occur on nightguards and removable orthodontic retainers. Care of these devices also includes daily rinsing/brushing and soaking when not in use.
Mouthrinses with antimicrobial properties can also be an effective tool in reducing the quantity of microorganisms causing halitosis. Products that have been proven to be effective usually contain active ingredients of chlorhexidine, essential oils, triclosan, or cetylpyridinium chloride.5 Since many halitosis conditions present specific and sometimes multiple etiologies, the proper prescription of an effective mouthrinse is different for each individual.
Research has shown a correlation between dry mouth and an increase of halitosis.17,18 Mouthrinses such as ACT® Dry Mouth (Chattem, Inc., www.chattem.com) or Biotene® (GlaxoSmithKine, www.gsk.com) are suggested for patients whose halitosis can be linked to an excessively dry mouth. The latter can be genetic or related to mouth breathing or medications taken. The medications include commonly used diuretics (eg, hydrochlorothiazide (Microzide), furosemide (Lasix)), psychiatric medications (eg, fluoxetine (Prozac), bupropion (Wellbutrin), paroxetine (Paxil)), some antibiotics (eg, metronidazole (Flagyl)), and asthma medications (eg, albuterol (Proventil)). Alternatively, patients with excessive amounts of saliva should use an alcohol-containing rinse such as Listerine® (Johnson & Johnson Healthcare, www.jnj.com/healthcare-products) or Scope® (The Procter & Gamble Company, www.pg.com).
A summary of the most commonly used chemical agents used for reducing halitosis has previously been discussed.9,11,19-25
Otolaryngeal Etiologies
When dental and periodontal etiologies are corrected or ruled out and oral malodor still persists, the next step is to consider sources from ear, nose, and/or throat (ENT) pathologies.10 Pharyngitis, tonsillitis, deep crypts of the tonsils that harbor bacteria, tonsilloliths, and/or sinusitis should all be evaluated as potential etiologies and the patient referred for evaluation and treatment to an ENT specialist.10
Systemic Etiologies
Beyond dental and ENT etiologies, other causes of halitosis can be traced to systemic conditions requiring consultation and treatment from a medical doctor. Bronchitis, bronchial carcinomas, and chronic airway obstruction can create foul malodor.10 Regurgitation esophagitis and helicobacter pylori infection of the stomach often cause a foul malodor.10 This is due to reflux of stomach acids into the esophagus and sometimes into the upper oral cavity. A patient presenting with a sweet or ammonia type form of halitosis may have liver cirrhosis or liver pathology.10
Kidney insufficiency sometimes manifests itself in the form of a fishy type odor. Patients with this condition may also complain about perpetually salty tasting saliva.10 A distinctive sweet smell or one of rotten apples may be reason to suggest a patient be tested for diabetes. The accumulation of ketone bodies associated with untreated diabetes is the cause of this odor.10 When a patient with halitosis presents with sweet smelling malodor, especially if there is a family history or the patient him or herself exhibits risk factors for diabetes, he or she should be instructed to have blood glucose levels evaluated. Certain medications may also be the source of particular malodors. Some antibiotics such as metronidazole can create a metallic smell. Eucalyptus-containing medications can create a rotting melon smell. Arsenic can cause an onion-like odor.10 If the source of malodor is a particular medication, possible non-odor–causing substitutions are advised, if possible.
Social Etiologies
A patient’s diet is another factor that should be discussed when recommending a plan to combat oral malodor. Frequent consumption of spicy foods, onions, garlic, dairy, or alcohol may result in unpleasant odors and halitosis. The dentist should suggest that the patient keep a journal of his or her weekly food intake and also experiment with the removal of the aforementioned items from their diets to identify the foods responsible for the offending odors.
In conjunction with suggestion of eliminating foods shown to leave lingering malodor, the suggestion of regular intake of probiotics has shown to be beneficial in treating halitosis.11 This practice can help reduce VSC-producing bacteria found in both the oral cavity and gut. Studies have shown that probiotic bacterial strains sourced from indigenous oral microbiotas of healthy humans may potentially aid in prevention and treatment of halitosis.19 Emergent halitosis treatment research includes probiotics and vaccines targeted against these specific causal microorganisms or their products.20
Smoking is a habit that can also contribute to halitosis. Tar and nicotine build-up becomes increasingly difficult to mask in heavy and long-term smokers. Smoking cessation is advised in these cases once adjunctive respiratory pathologies are ruled out by a physician. Smoking cessation can include prescribing products such as Chantix® (varenicline) (Pfizer, www.pfizerpro.com) or Wellbutrin® (bupropion) (GlaxoSmithKline), hypnosis therapy, and use of smokeless cigarettes.
When considering social etiologies, it is also important to realize that while an odor might cause a clinician to make a diagnosis of halitosis, the odor may not seem offensive to the individual patient. For example, certain wines or liquors may produce a distinctive odor, not identified as halitosis by the patient but objectionable by social contacts. In certain individuals, “garlic breath” due to an intake of garlic-containing foods or indigenous to a culture may not be considered to be malodor by the people close to the individual. If the odor is negatively affecting the patient’s social interactions, he or she will be more likely to present and request treatment. The following cases demonstrate how determination of etiology and individual treatment protocol was effective in treating patients with a chief complaint of halitosis.
Case 1
A 27-year-old Caucasian man presented to the office with a chief complaint of having a constant bad smell in his mouth that he said had been getting worse over the past few months. He noticed that close friends told him they were also aware of his bad breath. The patient had a non-remarkable medical history. His past dental treatment included numerous dental restorations, and his last professional hygiene appointment was approximately 2 years prior. The patient was a non-smoker, drank alcohol on social occasions, and did not report a diet that included large volumes of onions, garlic, or spicy foods.
Evaluation of the patient’s full mouth series of radiographs revealed signs of mild periodontal bone loss and a large carious lesion/defective restoration on tooth No. 19. A treatment plan was devised that included oral hygiene instructions, scaling/root planing, root canal treatment on No. 19, crown fabrication on No. 19, and re-evaluation 4 to 6 weeks later. During the re-evaluation appointment, oral hygiene was shown to be slightly improved, and a 3-month periodontal maintenance interval was suggested (Figure 8). The patient reported a significantly decreased degree of malodor and was very satisfied.
Case 2
A 46-year-old Hispanic woman presented to the office stating the following: “I have terrible breath that smells like rotting eggs that is especially strong in the morning.” Her medical history was non-remarkable, and she reported having a professional dental cleaning 6 months prior. The patient was a non-smoker, non-drinker, and reported being very fond of onions. Evaluation of the oral cavity revealed no caries or periodontal inflammation, but the large size of the patient’s tonsils was noted (Figure 9).
The treatment for the patient included oral hygiene instructions and full-mouth scaling and root planing. The patient was then instructed to make the following lifestyle/hygiene adjustments for a week at a time prior to her 1-month re-evaluation appointment:
• Week 1: Eliminate all onions, garlic, and spicy foods from the diet.
• Week 2: Rinse/gargle twice a day with 0.12% chlorhexidine rinse.
• Week 3: Rinse/gargle twice a day with a 1.5% hydrogen peroxide rinse
The patient was instructed to keep a daily journal describing her perception of the oral malodor at the end of each of these 3 weeks. In addition, the patient was referred to an ENT specialist to evaluate her tonsils as the possible etiology. The patient was diagnosed as having deep tonsilar crypts in which food debris and bacteria were being lodged. Discussion between the dental team and the medical doctor concluded that if neither the chlorhexidine nor hydrogen peroxide rinses were effective in keeping the tonsilar area clean, surgical removal of the tonsils would be discussed with the patient as a treatment option.
During the patient’s re-evaluation appointment a month after her initial visit, the patient reported the following observations from her journal: the week of eliminating odorous foods from her diet did not resolve malodor; she noticed improvement with use of the chlorhexidine rinses but complained of the brown residual staining left on her teeth as a result; improvement was also noted with use of the hydrogen peroxide rinse. These were evidence that the tonsilar crypts were retaining food and bacteria and were the etiology of her halitosis.
After discussing the findings with the ENT doctor along with these outcomes, it was determined that the hydrogen peroxide rinse should be incorporated into her daily home hygiene regimen, and surgical intervention was not needed at this time. The patient was and continues to date to be satisfied with this result.
Conclusions
After determining the etiology of the halitosis and then considering all the possible mechanical and chemical treatments available, an individualized treatment plan should be created by the dentist or dental hygienist. This plan should include consultation with the patient’s physician or specific medical specialists if deemed necessary. It is essential that the patient and dentist continue to monitor halitosis via regular re-evaluation and maintenance even after successful treatment. With the long-term goal being to eliminate offensive malodor, management is a process that requires regular professional monitoring, treatment, and effective home hygiene practices.
Halitosis is an embarrassing condition affecting many members of the adult population. With proper diagnosis, identification of the etiology, and timely referrals when needed, steps can be taken to create a successful individualized treatment approach for each patient seeking assistance. This can be an excellent source of patient referrals.
DISCLOSURE
The authors declare no affiliation with any of the companies mentioned in this article.
ABOUT THE AUTHORS
Stuart J. Froum, DDS
Clinical Professor and Director of Clinical Research, Department of Periodontology and Implant Dentistry,
New York University College of Dentistry,
New York, New York;
Diplomate, American Board of Periodontology;
Diplomate, International Congress of Oral Implantology,
Periodontics and Implant Dentistry
Kristina Rodriguez Salaverry, DDS, MS
Instructor of Clinical Dental Medicine,
Columbia University College of Dental Medicine,
New York, New York;
Diplomate, American Board of Periodontology
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
REFERENCES
1. Tonzetich J, Ng SK. Reduction of malodor by oral cleansing procedures. Oral Surg Oral Med Oral Pathol. 1976;42(2):172-181.
2. Bosy A. Oral malodor: philosophical and practical aspects. J Can Dent Assoc. 1997;63(3):196-201.
3. Ayers KM, Colquhoun AN. Halitosis: causes, diagnosis, and treatment. N Z Den J. 1998;9(418):156-160.
4. Attia EL, Marshall KG. Halitosis. Can Med Assoc J. 1982;126(11):1281-1285.
5. van den Broek AM, Feenstra L, de Baat C. A review of the current literature on management of halitosis. Oral Dis. 2008;14(1):30-39.
6. Cortelli JR, Barbosa MD, Westphal MA. Halitosis: a review of associated factors and therapeutic approach. Braz Oral Res. 2008;22(suppl 1):44-54.
7. Tonzetich J. Production and origin of oral malodor: a review of mechanisms and methods of analysis. J Periodontol. 1977;48(1):13-20.
8. Rosenberg M, McCulloch CA. Measurement of oral malodor: current methods and future prospects. J Periodontol. 1992;63(9):776-782.
9. Rosenberg M, Gelernter I, Barki M, Bar-Ness R. Daylong reduction of oral malodor by two-phase oil:water mouthrinse, as compared to chlorhexidine and placebo rinses. J Periodontol. 1992;63(1):39-43.
10. van Steenberghe D. Breath Malodor: A Step-by-step Approach. Copenhagen, Denmark: Quintessence Publishing; 2004.
11. Bollen CM, Beikler T. Halitosis: the multidisciplinary approach. Int J Oral Sci. 2012;4(2):55-63.
12. Rosenberg M, Septon I, Eli I, et al. Halitosis measurement by an industrial sulphide monitor. J Periodontol. 1991;62(8):487-489.
13. Preti G, Clark L, Cowart BJ, et al. Non-oral etiologies of oral malodor and altered chemosensation. J Periodontol. 1992;63(9):790-796.
14. Stammberger H. Endoscopic endonasal surgery—concepts in treatment of recurring rhinosinusitis. Part II. Surgical technique. Otolaryngol Head Neck Surg. 1986;94(2):47-56.
15. Poyato-Ferrera M, Segura-Egea JJ, Bullón-Fernández P. Comparison of modified Bass technique with normal toothbrushing practices for efficacy in supragingival plaque removal. Int J Dent Hyg. 2003;1(2):110-114.
16. Kaizu T, Tsunoda M, Aoki H, Kimura K. Analysis of volatile sulphur compounds in mouth air by gas chromatography. Bull Tokyo Dent Coll. 1978;19(1):43-52.
17. Kleinberg I, Wolff MS, Codipilly DM. Role of saliva in oral dryness, oral feel and oral malodour. Int Dent J. 2002;52(suppl 3):236-240.
18. Koshimune S, Awano S, Gohara K, et al. Low salivary flow and volatile sulfur compounds in mouth air. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003;96(1):38-41.
19. Pitts G, Brogdon C, Hu L, et al. Mechanism of action of an antiseptic, anti-odor mouthwash. J Dent Res. 1983;62(6):738-742.
20. Frascella J, Gilbert R, Fernandez P. Odor reduction potential of a chlorine dioxide mouthrinse. J Clin Dent. 1998;9(2):39-42.
21. Raven SJ, Matheson JR, Huntington E, Tonzetich J. The efficacy of a combined zinc and triclosan system in the prevention of oral malodour. In: van Steenberghe D, Rosenberg M, eds. Bad Breath: A Multidisciplinary Approach. Leuven, Belgium: Leuven University Press;1996:241-254.
22. Quirynen M, Zhao H, van Steenberghe D. Review of the treatment strategies for oral malodour. Clin Oral Investig. 2002;6(1):1-10.
23. Suarez FL, Furne JK, Springfield J, Levitt MD. Morning breath odor: influence of treatments on sulfur gases. J Dent Res. 2000;79(10):1773-1777.
24. Burton JP, Chilcott CN, Moore CJ, et al. A preliminary study of the effect of probiotic Streptococcus salivarius K12 on oral malodour parameters. J Appl Microbiol. 2006;100(4):754-764.
25. Scully C, Greenman J. Halitology (breath odour: aetiopathogenesis and management). Oral Dis. 2012;18(4):333-345.